ALERT!
This site is not optimized for Internet Explorer 8 (or older).
Please upgrade to a newer version of Internet Explorer or use an alternate browser such as Chrome or Firefox.
Segmentectomies Made Easy: Robotic-Assisted Left S8 Segmentectomy
Mouyer Z, Mughal A, Elfiky A, M. Habib A. Segmentectomies Made Easy: Robotic-Assisted Left S8 Segmentectomy. February 2026. doi:10.25373/ctsnet.31390806
This video is part of CTSNet’s 2025 Resident Video Competition. Watch all entries into the competition, including the winning videos.
Robotic-assisted thoracic surgery (RATS) has emerged as an invaluable tool in the management of early-stage lung malignancies compared to traditional video-assisted thoracoscopic surgery (VATS), offering improved operative precision, dexterity, and three-dimensional (3D) visualization. A segmentectomy in a particularly anatomically complex or deep segment, such as S8, remains a technically demanding task. Recent advances in 3D imaging technologies have transformed preoperative planning and intraoperative guidance (1).
3D computed tomography (CT) reconstructions have been shown to enhance anatomical accuracy and operative efficiency in robotic segmentectomy (1). In robotic-assisted lung resections, 3D modelling enables more precise visualization of anatomical relationships and has been shown to improve intraoperative orientation, operator confidence, reduced open conversion rate, and dissection accuracy. A 2024 prospective trial reported that 3D virtual lung reconstruction led to a significant increase in confidence in surgical plans and a low conversion rate to lobectomy (8.96 percent) in robotic segmentectomy (2). Initial studies suggest that there is no significant difference between RATS and VATS (3). Nevertheless, compared to VATS, RATS is still a novel technique, with few expert veteran operators. As RATS is adopted more widely among thoracic centers globally, there is an increasing need to continually re-evaluate clinical outcomes and compare them with established techniques such as VATS. At the authors’ institution, they have achieved significant success with RATS and continue to refine their surgical approach through cumulative experience and ongoing learning.
In this video tutorial, the authors present a robotic S8 segmentectomy for a deep pulmonary metastasis, uniquely guided by high-resolution 3D reconstruction imaging. This case exemplifies how advanced 3D visualization can enhance surgical planning by optimizing dissection strategy and minimizing unnecessary parenchymal disruption.
Patient Presentation and Preoperative imaging (0:13)
A 74-year-old female patient who was a former smoker with a history of breast cancer in remission presented to the thoracic surgery service (Figure 1) with a suspicious lung nodule. Further diagnostic imaging identified a 12 mm lesion in the S8 segment of the left lower lobe (Figure 2). Preoperative lung function showed both a forced expiratory volume in one second of 100 percent and a diffusing capacity of the lung for carbon monoxide of 104 percent. She had no other significant past medical history. The patient had a World Health Organization performance status of one and an American Society of Anesthesiology grade class two.
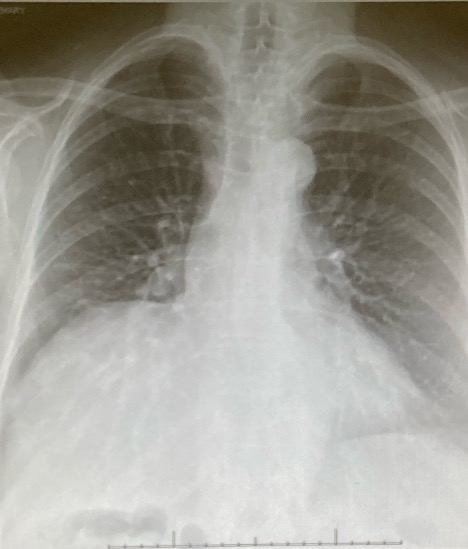
Figure 1: An image of the preoperative chest X-ray (CXR).

Figure 2: Images of the preoperative CT scans in the axial and coronal planes.
The patient underwent positron emission tomography (PET)/CT imaging that showed a fluorodeoxyglucose-avid lesion in the right upper lobe, yielding a Herder score of 91.7 percent, suggesting a very high likelihood of malignancy (Figures 3, 4). Following a multidisciplinary team discussion, the patient was subsequently recommended for a robotic-assisted segmental resection of the left S8 segment. This procedure was performed using a robotic system.

Figure 3: An image of the axial positron emission tomography (PET)/CT scan.
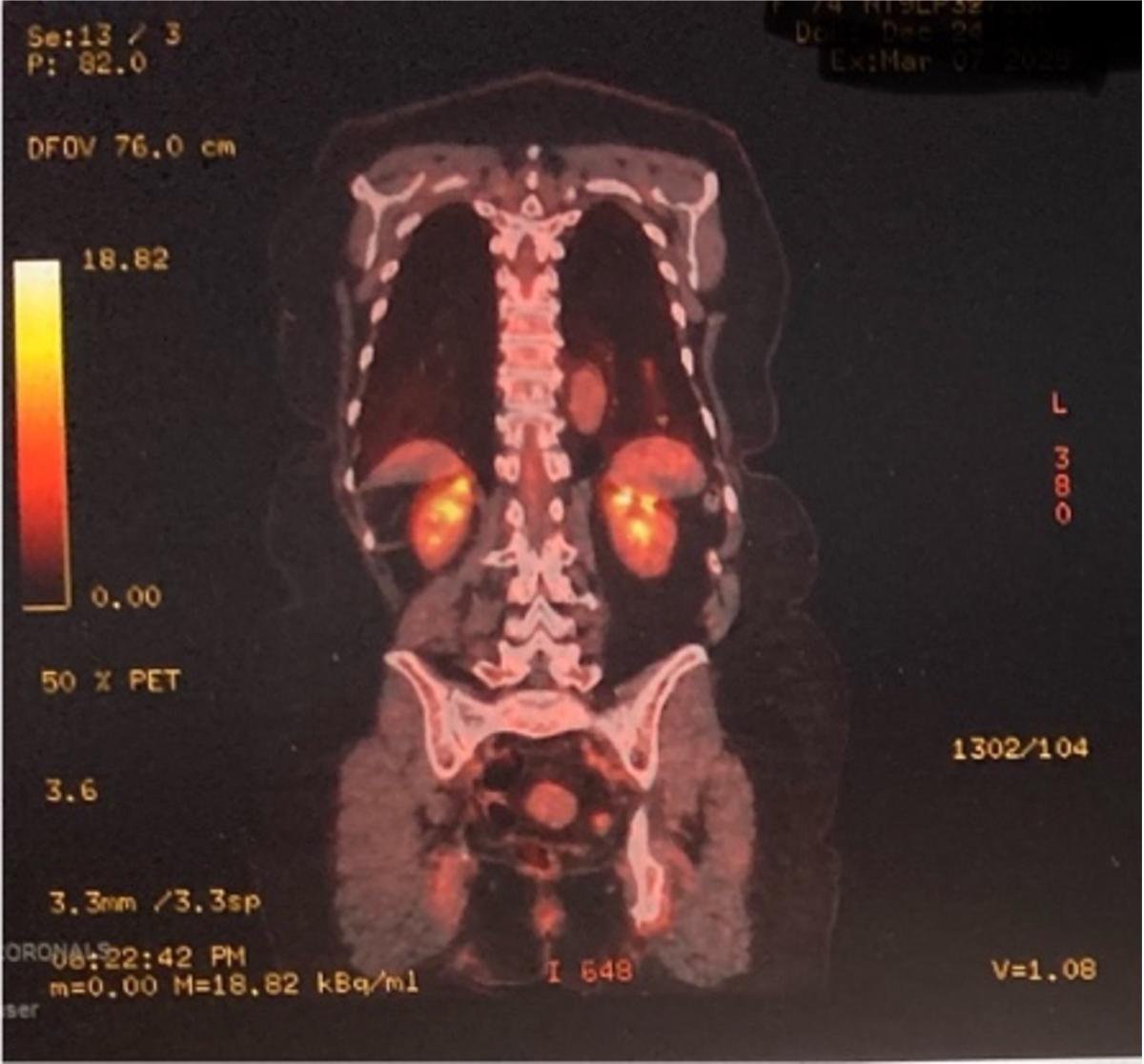
Figure 4: An image of the PET/CT coronal scan.
The CT images revealed a 12 mm spiculated nodule in the S8 segment of the left lower lobe (Figures 1, 2).
The preoperative workup revealed a forced expiratory volume in one second of 109 percent and a diffusing capacity of the lung for carbon monoxide of 100 percent. The patient had a World Health Organization performance status of one and an American Society of Anesthesiology grade class three.
The PET/CT imaging showed a fluorodeoxyglucose-avid lesion in the left lower lobe and a Herder score of 91.7 percent (Figure 3).
Following the multidisciplinary team discussion, the patient was subsequently recommended for a robotic-assisted segmental resection of the left S8 segment.
Utilization of 3D Reconstruction to Optimize the Segment 8 Segmentectomy Approach in the Left Lower Lobe (0:54)
3D reconstructive imaging was employed using the Innersight platform based on CT data from the patient. This approach enabled highly detailed anatomical visualization and preoperative planning. The initial reconstruction focused on the lateral and inferior aspects of the left lung, delineating the left lower lobe and its segments S6a, S6b, S7a, S7b, S8 and S9.
Upon selectively removing the S8 parenchyma, a 12 mm lesion became visible, along with the surrounding segmental vasculature and airways. The conventional surgical technique for S8 segmentectomy typically involves a posterior approach through the oblique fissure to access the left main pulmonary artery. Upon entering the fissure, the arterial branch to the apical S6 of the left lower lobe was identified and served as a key anatomical landmark to guide dissection inferiorly through the inferior parenchyma.
To demonstrate the depth of parenchyma that would need to be dissected with this method, both S6a and S6b were virtually removed. Identification and dissection along the lingular artery then enabled visualization of the common basilar trunk, through which the segmental artery to S8 was subsequently found. This approach represents the conventional strategy widely described in the literature and was the initial operative plan.
However, through iterative review of the 3D reconstruction, a novel and minimally disruptive route that entirely avoided deep parenchymal dissection was identified. By restoring the full anatomical model with transparent segment overlays, an inferior approach along the inferior pulmonary vein was explored. Upon resecting S8 inferiorly in the simulation, four venous tributaries draining into the inferior pulmonary vein were observed. Notably, the bronchus to S8 was clearly identifiable between the two most inferior tributaries, with the S8 artery positioned centrally posterior to the S8 bronchus.
Intraoperatively, this anatomical insight allowed for precise identification and stapling of the S8 bronchus, followed by stapling of the S8 artery, all with no dissection of the surrounding lung parenchyma. Compared to the standard posterolateral approach, this inferior route proved to be less invasive and potentially superior.
Preoperative Setup and Initial Dissection (3:44)
The surgical procedure was performed using the a robotic platform with the following instruments: forceps, bipolar fenestrated forceps, a bipolar electrosurgical instrument, and a monopolar hook. Upon entering the thoracic cavity, an initial assessment of the oblique fissure was performed to evaluate completeness and to identify any pleural adhesions.
The lung was then retracted anteriorly to expose the posterior basal segments. Upon inspection, the lesion in segment S8 was localized by visualizing subtle puckering and palpating firm resistance in the inferior portion of the left lower lobe. The suspected location of the S8 lesion was marked using diathermy for subsequent orientation.
Following lesion localization, the lung was repositioned superiorly to allow for inferior access. The inferior pulmonary vein was then identified, and its four segmental tributaries were clearly visualized, in accordance with the preoperative 3D reconstruction. Notably, this exposure was achieved without the need for any parenchymal dissection, thereby preserving lung tissue. In contrast, a conventional lateral or posterior approach via the oblique fissure would have necessitated deep dissection through the thickest portion of the lung parenchyma to reach the same anatomical targets.
Dissection was performed using a bipolar electrosurgical instrument for precise and controlled division of tissues. Inferior to the S8 bronchus and directly within the operative field, the station 12 lymph nodes were excised. This action not only aided in exposure but also ensured oncological completeness.
Stapling of the S8 Bronchus (6:05)
The S8 bronchus was clearly visualized, with the S8 arterial branch identified coursing directly beneath it. The bronchus was then captured and stapled using a thick tissue stapler load.
Stapling of the S8 Artery (6:20)
The S8 artery was carefully dissected, captured, and stapled with a vascular staple load.
Injection of Indocyanine Green, Pyramidal Parenchymal Stapling, and Removal of the Specimen (6:54)
Infrared fluorescence imaging was employed following the intravenous injection of indocyanine green (ICG). The demarcation of the isolated S8 was sharply defined, closely mirroring the preoperative 3D reconstruction. Using a monopolar diathermy hook, the intersegmental borders were scored, ensuring adequate oncological margins around the nodule. The infrared filter was then removed, and the S8 segment was resected along the marked lines using sequential stapling. Special attention was paid to gradual anterior and posterior stapling towards the apex of the segmental pyramid to prevent distortion or folding of adjacent basal segments, thus preserving postoperative parenchymal inflation. Once the segmental apex was reached, the remaining parenchyma was minimal and was easily stapled across for completion of the resection. The remaining vascular and airway stumps were seen without obstruction.
Once the parenchymal stapling was completed, the specimens were placed in a specimen retrieval bag and removed from the chest. Before closure, a chest drain was inserted, and inflation of the remaining upper lobe segments was confirmed.
Postoperative Course and Histological Analysis (9:32)
Postoperatively, the chest drain was removed at six hours (Figure 5), and the patient was discharged 20 hours after the operation on postoperative day one (Figure 6). The patient experienced no postoperative complications and had an uneventful postoperative recovery.
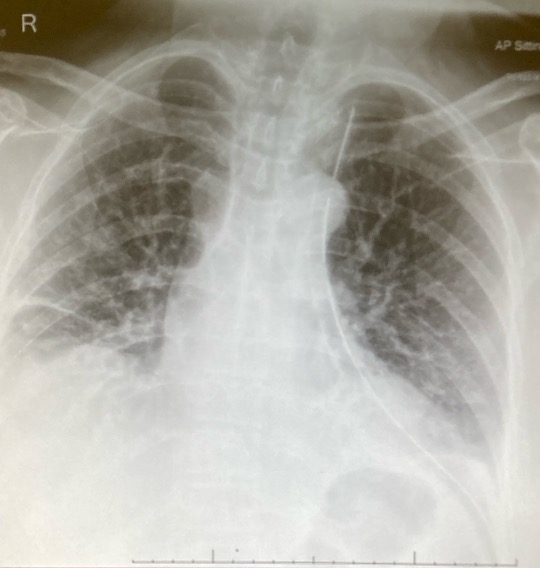
Figure 5: An image of the postoperative CXR with the drain in.
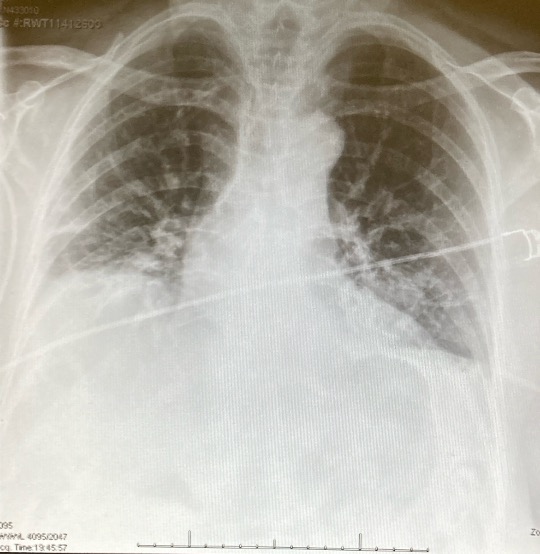
Figure 6: An image of the postoperative CXR with the drain out.
Histopathological analysis confirmed the lesion as a metastatic deposit from the patient’s primary breast adenocarcinoma, which was diagnosed 11 years prior. Complete resection with clear margins was achieved.
Outcome
The chest drain was removed six hours postoperatively, and the patient was discharged within 20 hours on postoperative day one. Histopathological analysis confirmed the lesion to be a completely resected metastatic breast adenocarcinoma, originating from the patient’s primary breast disease diagnosed 11 years prior. Resection margins were clear, and no further pulmonary lesions were identified. The patient was referred back to her breast oncology team for ongoing management and surveillance.
Discussion
This video tutorial is the first entry in the “Segmentectomy Made Easy” video atlas. This particular tutorial presents a robotic S8 segmentectomy for a deep pulmonary metastasis, uniquely guided by high-resolution 3D reconstruction imaging. This case exemplifies how advanced 3D visualization can enhance surgical planning by optimizing dissection strategies, mapping any atypical vasculature or airways, and minimizing unnecessary parenchymal disruption.
Evidence in the literature on the application of 3D technology in minimally invasive thoracic surgery (VATS or RATS) is scarce. Nevertheless, Li et al. highlighted that a RATS S7 and S8 segmentectomy is safe and feasible with the utilization of 3D reconstruction because it enables better identification of segmental structures and facilitates efficient dissection (4).
Promising ventures include the application of artificial intelligence-assisted 3D reconstruction in the preoperative planning process. Song et al. emphasized that artificial intelligence-assisted 3D reconstruction significantly reduced reconstruction time and improved the accuracy of sublobar localization of pulmonary lesions, particularly those at segment junctions (5). Additionally, Ong et al. demonstrated the utility of preoperative 3D reconstruction in guiding thoracoscopic S8 segmentectomy, emphasizing its role in overcoming the multiple and varying anatomical challenges (6).
Despite these promising developments, further research is necessary to establish standardized protocols and assess long-term outcomes associated with the integration of 3D reconstruction technologies in RATS. The authors hope video series like these act as a step in the right direction in developing the standard protocol needed to successfully perform 3D-guided RATS segmentectomies. Ongoing studies, such as the DRIVATS trial (3D computed tomography reconstruction in video-assisted thoracoscopic segmentectomy), are designed to evaluate the routine use of 3D reconstruction for lung resections, particularly segmentectomies, to confirm their value in improving surgical outcomes (7).
The incorporation of 3D reconstruction technologies into RATS has enhanced the precision and safety of segmentectomies, especially in challenging segments such as S8. These advances contribute to improved surgical planning, reduced conversion rates, and potentially better postoperative outcomes. Continued research and technological refinement are essential to fully realize the beneficial potential of these innovations in thoracic surgery.
References
- Eguchi T, Sato T, Shimizu K. Technical advances in segmentectomy for lung cancer: a minimally invasive strategy for deep, small, and impalpable tumors. Cancers (Basel) 2021;13:3137.
- Browne IL, Patel YS, Hanna NM, Haider E, Hanna WC. Three-dimensional virtual lung reconstruction in robotic segmentectomy: a safety and feasibility trial. J Thorac Cardiovasc Surg Tech 2024;15:158–66.
- Tupper HI, Lawson BL, Kipnis P, Patel AR, Ashiku SK, Roubinian NH et al. Video-assisted vs robotic-assisted lung lobectomies for operating room resource utilization and patient outcomes. JAMA Netw Open 2024;7:e248881.
- Li JT, Huang J, Luo QQ. Robotic-assisted right medial and anterior basal segmentectomy (S7+S8). J Thorac Dis 2018;10:E562–6.
- Song Z, Izhar A, Wei B. Artificial intelligence-assisted three-dimensional reconstruction in thoracic surgery: a narrative review. Current Challenges in Thoracic Surgery 2025;7:15.
- Ong CW. Video-assisted thoracoscopic left S8 segmentectomy guided by pre-operative 3D reconstruction in a patient with synchronous bilateral primary lung cancer: a case report. J Vis Surg 2023;9:45.
- Niu Z, Chen K, Jin R, Zheng B, Gong X, Nie Q et al. Three-dimensional computed tomography reconstruction in video-assisted thoracoscopic segmentectomy (DRIVATS): a prospective, multicenter randomized controlled trial. Front Surg 2022;9:941582.
Disclaimer
The information and views presented on CTSNet.org represent the views of the authors and contributors of the material and not of CTSNet. Please review our full disclaimer page here.




